Talk Overview
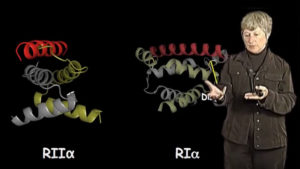
In this lecture, I have given an overview of protein kinase structure and function using cyclic AMP dependent kinase (PKA) as a prototype for this enzyme superfamily. I have demonstrated what we have learned from the overall structural kinome which allows us to compare many protein kinases and also to appreciate how the highly regulated eukaryotic protein kinase has evolved. By comparing many protein kinase structures, we are beginning to elucidate general rules of architecture. In addition, I have attempted to illustrate how PKA is regulated by cAMP and how it is localized to specific macromolecular complexes through scaffold proteins.
Speaker Bio
Susan Taylor

Dr. Taylor received her BA in Chemistry from the University of Wisconsin-Madison and her PhD in Physiological Chemistry from Johns Hopkins University. After completing a fellowship at the MRC in Cambridge, she moved to the University of California, San Diego where she soon secured a faculty position in the chemistry department. Shortly after joining UCSD,… Continue Reading







india parker says
Hi Susan! Thank you so much for teaching me:]