Talk Overview
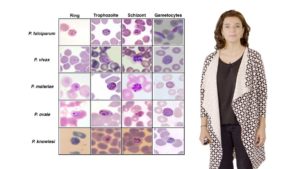
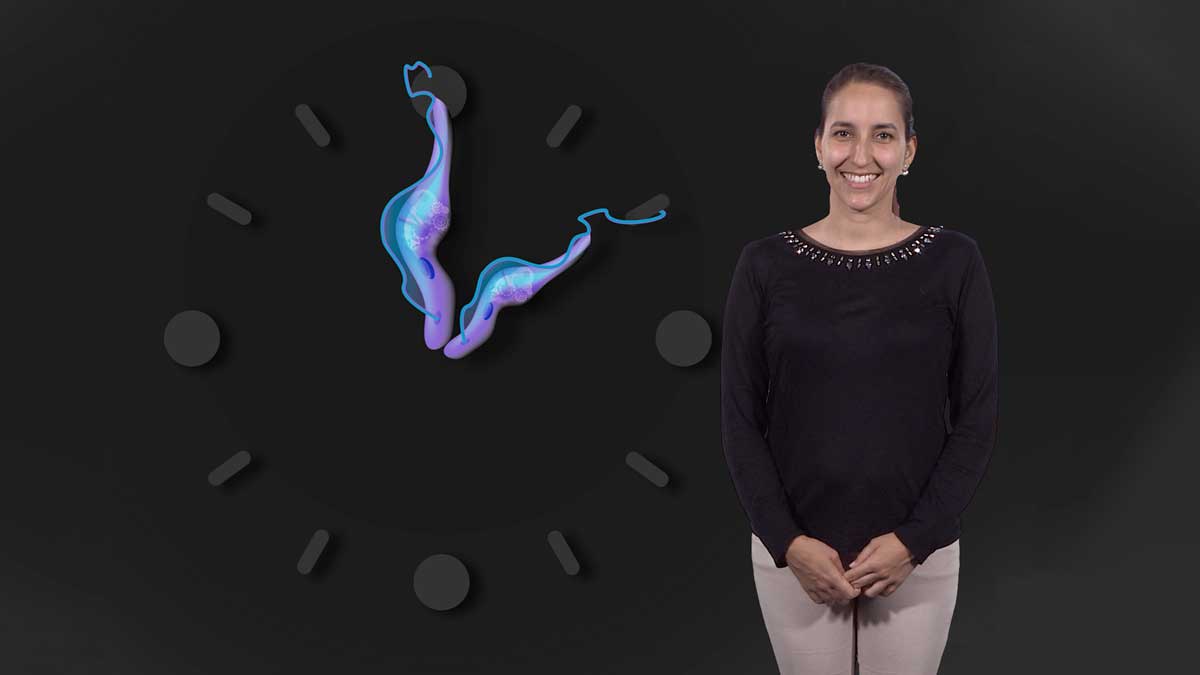
Malaria is currently responsible for about 500,000 deaths per year and is especially fatal to children under the age of 5 years. Two global eradication programs since 1950 have reduced the malaria burden significantly, however, progress has stalled in recent years. In her first talk, Dr. Maria Mota details the lifecycle of two malaria parasites, Plasmodium falciparum and P. vivax. The complex, multistage lifecycle makes it hard to diagnose and treat malaria. Mota explains that many attempts have been made to treat the disease with drugs but the malaria parasite very rapidly develops resistance. She describes how the recent development of drug combinations and a new vaccine, together with insecticide treated bed nets and improved early diagnosis, provide hope for better prevention and treatment of malaria in the future.
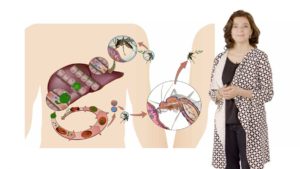
In Part 2, Mota goes into more depth about the liver stage of malaria infection. She reminds us that when Plasmodium parasites are transferred by a mosquito bite, they first travel to the liver. Once inside of a hepatocyte, a single malaria parasite will replicate and give rise to over 10,000 new parasites that go on to infect red blood cells and cause disease. For many years, this stage was called the silent stage as it was thought that malaria parasites were not detected by the host while in the liver. Mota tells us about data from her lab, and others, showing that the host innate immune response is, indeed, activated during hepatocyte infection. This immune response may play a role in modulating the host’s response to the blood-stage infection or future re-infection.
The severity of malarial disease is influenced by interactions between the parasite, the host and environmental factors. In her third talk, Mota explains how her lab used a mouse model to study the impact of host nutritional status on disease severity. When they compared infections in calorically restricted (CR) mice and freely fed mice, they found less reproduction of the malaria parasite and a lower parasite load in CR mice. This suggested that Plasmodium are able to sense and respond to host nutritional status. Mota describes how her lab identified a key Plasmodium kinase, KIN, that seems to regulate parasite response to nutrient availability and may provide a target for antimalarial drugs.
Speaker Bio
Maria Mota

Dr. Maria Mota has been a Group Leader at the Instituto de Medicina Molecular (iMM) in Lisbon, Portugal since 2005 and Executive Director since 2014. She is also an invited Professor in the Faculty of Medicine at the University of Lisbon. Her lab studies the complex interactions between the malaria parasite Plasmodium and the human… Continue Reading








Moses Bulamu says
That’s so fabulous. I would like to be part of the science group of researchers please mail me now