Talk Overview
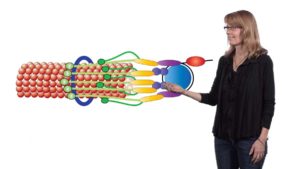
Proper chromosome segregation during cell division is critical to ensure that daughter cells inherit the correct number of chromosomes. Microtubules emanating from the spindle poles pull on sister chromatids to move one chromosome to each pole. The kinetochore, a protein complex on the chromosome, is key to regulating chromosome segregation. Kinetochores form attachments to microtubule ends (no easy feat since microtubules are constantly growing and shrinking), they sense tension to ensure that sister chromatids are connected to microtubules from opposite poles, and they signal the cell to stop cell division if attachment is not correct. Biggins gives an excellent overview of kinetochore structure and its critical functions in chromosome segregation.
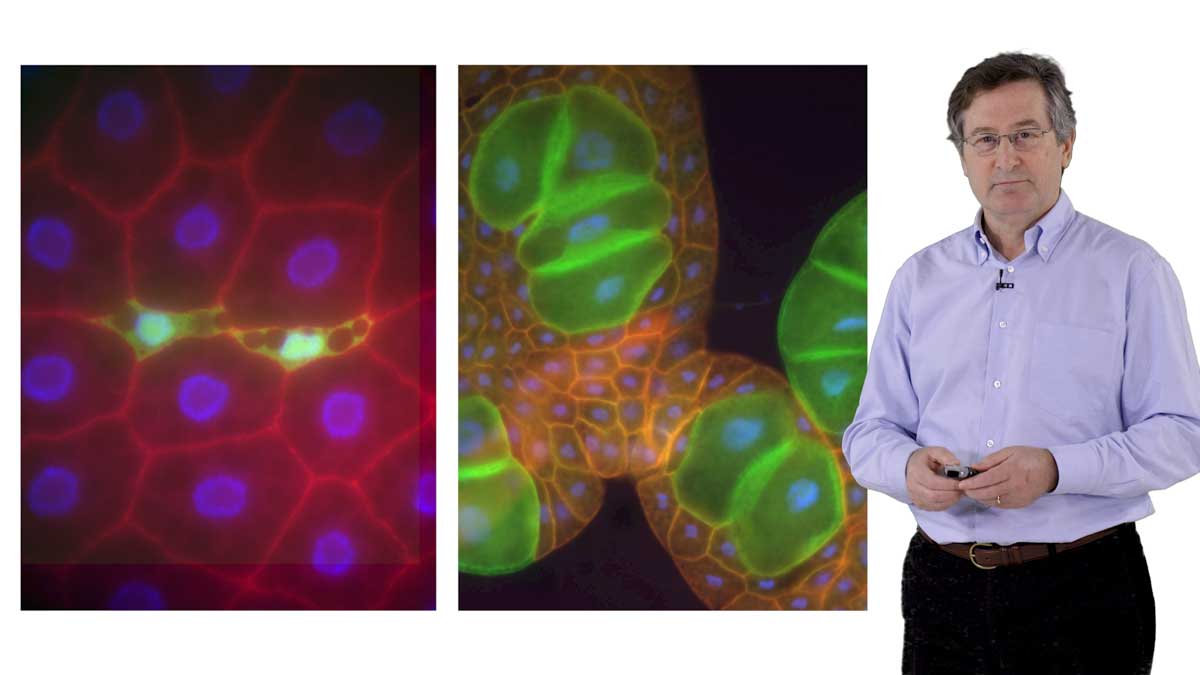
When Biggins began working on kinetochores, the experiments that she could do were limited by the lack of a method to purify intact kinetochores. In Part 2 of her talk, Biggins explains how her lab purified kinetochores from yeast (for the first time ever!). They showed that the purified protein complex functioned in the same manner in vitro as endogenous kinetochores. Using electron microscopy and other techniques, Biggins and her collaborators were able to visualize the structure of the kinetochore-microtubule attachment and demonstrate, surprisingly, that tension directly stabilizes the attachment.
Speaker Bio
Sue Biggins

Dr. Sue Biggins studied biology as an undergraduate at Stanford University and initially thought she would apply to medical school after receiving her degree. However, after a summer working in a research lab, she changed her mind and decided to apply to graduate school. Biggins received her PhD in molecular biology from Princeton and was… Continue Reading






pankaj rathour says
thank you, so much.