Talk Overview
Yednock relates the discovery and development, over 15 years, of the drug Tysabri, an alpha4 integrin antibody, as a treatment for multiple sclerosis.
In the first of his two talks, Ted Yednock begins with an overview of multiple sclerosis. He describes how, in MS, immune cells are able to transverse the wall of blood vessels and infiltrate the brain and central nervous system resulting in damage to the myelin surrounding neurons. Yednock and his colleagues hypothesized that by blocking the infiltration of immune cells into the brain, the progression of the disease might be slowed. They went on to identify alpha4 integrin as the molecule that mediates adhesion of immune cells to the blood vessel wall, and they found that an alpha4 integrin antibody (Natalizumab or Tysabri) could block infiltration of the blood cells into the brain in a animal model of MS. Yednock then details the clinical development of Tysabri through to its approval by the FDA in 2004.
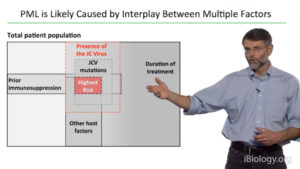
Just a few months after Tysabri was approved for MS treatment, two patients developed progressive multifocal leukoencephalopathy (PML), a fatal or seriously debilitating disease. In his second talk, Yednock describes the response of medical and regulatory groups and researchers to this discovery and its impact on the treatment of MS patients with Natalizumab/Tysabri.
Speaker Bio
Ted Yednock

Dr. Yednock recently joined the Drug Advisory Board of the Myelin Repair Foundation, a nonprofit organization focused on the discovery and development of therapeutics for multiple sclerosis (MS). Previously Dr. Yednock was the Executive Vice President and Head of Global Research for Elan Pharmaceuticals and a scientist at Athena Neurosciences. Through his scientific work at… Continue Reading








Thomasoneni says
Buying Essays Online – What to Watch for: https://vk.cc/9OTGak?id=buyessayonline1126vj
Thomasoneni says
Buy Essays Canada – Cheap & Safe Online Writing Service: https://vk.cc/9OTGak?id=buyessayonline112ncf
Timothyjeaks says
Buy Essay Online At Writing Service from Australia: https://vk.cc/9OTGak?id=buyessayonline113sxc