Talk Overview
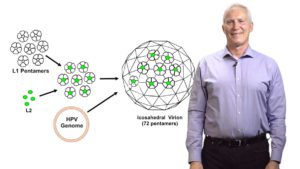
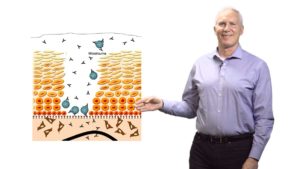
Human Papillomavirus (HPV) causes 5% of all cancers worldwide, and the first vaccine against HPV was approved in 2006. In this seminar, Dr. John Schiller provides an overview of HPV virus and infection, compares the three FDA approved vaccines against HPV, and explains the endpoints used in the clinical trials to prove vaccine efficacy. After a decade of using the vaccine, retrospective studies now allow us to evaluate the possibility of using single-dose vaccination, which could lead to an increase in the general use of the vaccine (implementation), and improve HPV-related cancer prevention.
In his second seminar, Schiller discusses the high efficacy of HPV vaccine, which is exceptionally good at producing neutralizing antibodies and also benefits from the low mutation rate of HPV. Coming to a better understanding of the efficacy of the HPV vaccine will provide evidence to support single-dose vaccination and aids in the development of new vaccines.
Speaker Bio
John Schiller

Dr. John Schiller is a National Institutes of Health (NIH) distinguished investigator and a professor at the National Cancer Institute at the NIH. Schiller completed his bachelor’s degree in molecular biology at the University of Wisconsin-Madison (1975), and received a doctorate degree in Microbiology at the University of Washington in Seattle (1982). Schiller continued his… Continue Reading




Becky says
“Human Papillomavirus (HPV) causes 5% of all cancers worldwide” … Really? I would appreciate the research that showed this. I have heard differently from someone who worked on the HPV vaccine. The marketing strategy included dropping some zeros and shifting a decimal or two to create the illusion of this figure. Also, the HPV causes specific cancers, not all…. and they differ between men and women. “Also, “the high efficacy of HPV vaccine” is a purely projected prognosis and there is as yet no appreciable data to prove that statement fully. In fact there is more data cropping up each month, suggesting that the vaccine has inadvertently caused some ovarian, colon and prostate cancers and rendered many women barren as well has having had a side effect of paralysis in varying degrees to young women the world over. While I understand how it is meant to work, that the theory and rationale is perfect, we have seen too many other variables, and in the light of the sheer magnitude of highly qualified whistleblowers who have specifically condemned this vaccine, it would be pertinent for parents to do a lot more research on how it has behaved in reality. I have some serious reservations about the air of marketing in this ‘biology’ lecture…. I would appreciate some bone fida clarity on this. Thanks 🙂
Erik says
Here’s the research that showed that:
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5520228/
Did you even watch the lectures?