Talk Overview
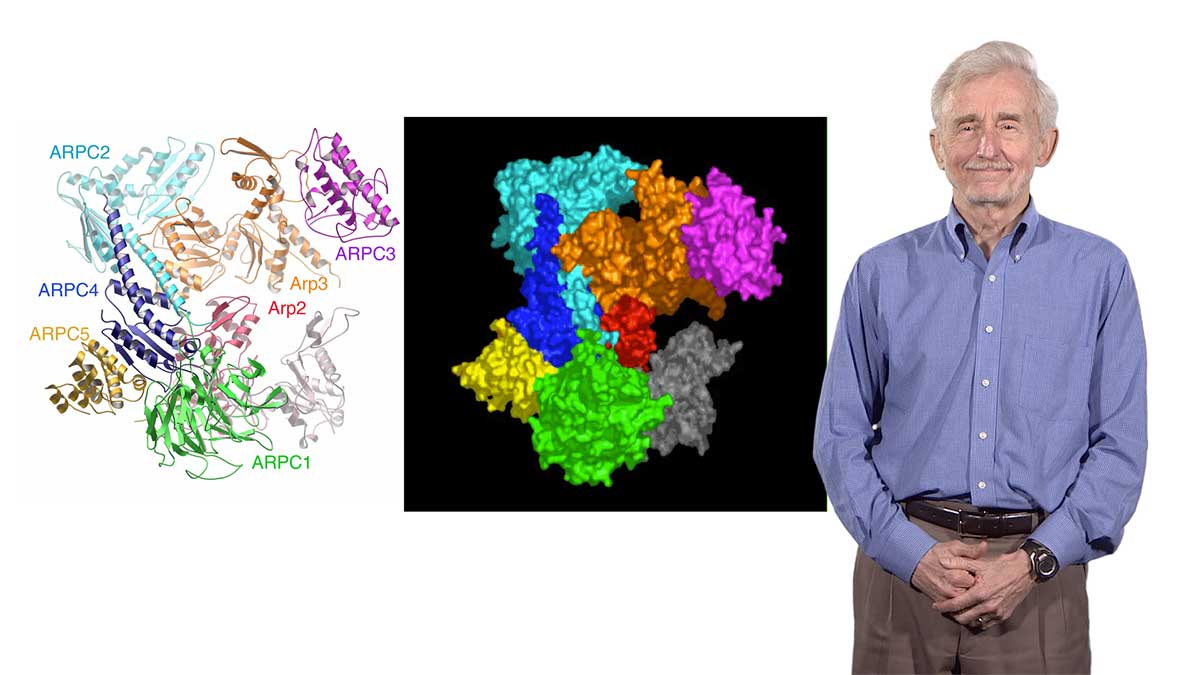
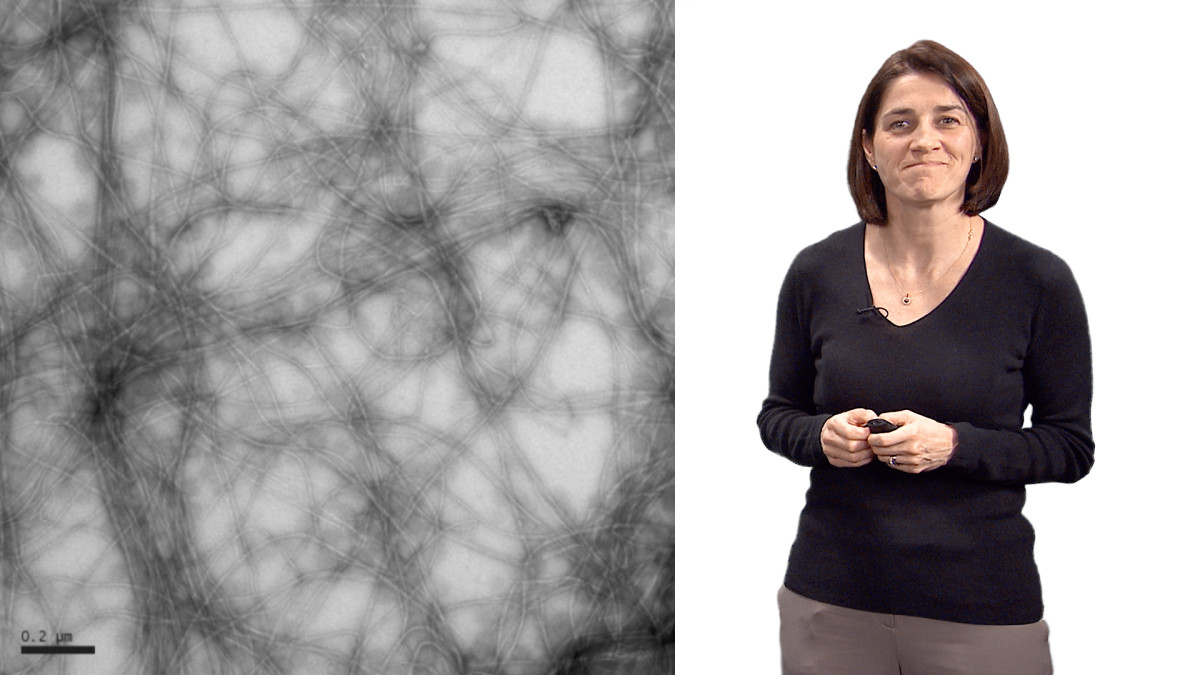
In Part 1 of her video, Dr. Theriot explains how tiny, nanometer sized actin molecules can self-assemble into filaments that are hundreds of microns in length. These actin filaments are constantly growing and shrinking, and this dynamic behavior allows a network of actin to generate enough force for cell motility. The intracellular bacterial pathogen Listeria monocytogenes uses actin polymerization to propel itself through the cytoplasm and to invade other cells. Many years of studies using Listeria have allowed Theriot and others to dissect the regulation of actin network growth in Listeria “comet tails” and at the leading edge of crawling cells.
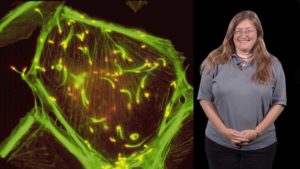
In her second lecture, Theriot explains that fish keratocytes are an excellent system to study rapid cell motility. By labeling actin and myosin in keratocytes, Theriot and her colleagues were able to follow turnover of actin in the lamellipodium. Unexpectedly, they found that myosin II plays in important role in actin disassembly at the rear of the cell and asymmetric localization of myosin at the back of the cell appears to govern cell turning. Similar mechanisms of actin and myosin cooperation seem to drive rapid movement in both fish keratinocytes and human neutrophils.
It has been known since the early 1990s that bacteria have homologues of both actin and tubulin. In general, however, bacteria are much simpler than eukaryotes. In her final lecture, Theriot speculates about the factors that have evolved to allow eukaryotes to modify their cytoskeletons and build bigger, morphologically complex, multicellular organisms.
Speaker Bio
Julie Theriot

Julie Theriot attended college at the Massachusetts Institute of Technology, graduating with degrees in Physics and Biology. She pursued graduate training at the University of California, San Francisco, earning her Ph.D. in Cell Biology in 1993. After four years as a Fellow at the Whitehead Institute for Biomedical Research, Theriot moved to Stanford University School… Continue Reading








Leave a Reply