Talk Overview
There are more than 5000 species of single-celled eukaryotes in the biological phylum known as the Apicomplexa, including the parasites responsible for malaria, neurological birth defects, and opportunistic infections associated with HIV/AIDS. These ancient protozoa provide a unique window into the evolution of subcellular organelles that have long fascinated cell biologists. Familiar features help to elucidate the origins, functions and design parameters for the secretory pathway, endosymbiotic organelles, the cytoskeleton, and cell cycle control. Conversely, parasite-specific organelles highlight the evolutionary diversity of eukaryotes, and suggest novel targets for treating disease.
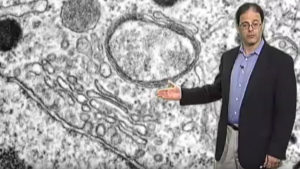
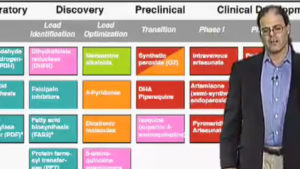
Antibiotics are effective because they kill bacteria without harming humans and other eukaryotes (organisms with cells that contain nuclei). So why are the eukaryotic parasites responsible for malaria and toxoplasmosis killed by drugs like clindamycin? Multidisciplinary studies integrating molecular genetics, cell biology, biochemistry, pharmacology and computational genomics reveal that such drugs target an unusual organelle. The “apicoplast” was acquired when an ancestral organism ‘ate’ a eukaryotic alga, and retained the algal plastid — a relative of plant chloroplasts derived from a bacterial ancestor. Although no longer photosynthetic, the apicoplast is essential for parasite survival, providing new targets for drug development.
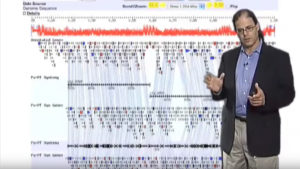
With the emergence of genomic-scale datasets representing all of the genes in the genome, all of the proteins in a cell or tissue, and all of the interactions and signals in an organism, biologists are increasingly faced with the challenge of how to store, integrate, and interrogate this information. How can we effectively mine large-scale datasets to expedite biological discovery, for example in the identification of new targets for anti-parasitic drug and vaccine design? Computational biology and genome informatics provide tools of growing importance for all biologists. Internet-based access makes such information available to scientists and the interested public worldwide.
Speaker Bio
David Roos

David S. Roos is the E. Otis Kendall Professor of Biology at the University of Pennsylvania, and Founding Director of the Penn Genomics Institute. He earned his undergraduate degree at Harvard College, a PhD at The Rockefeller University, and joined the University of Pennsylvania in 1989 after a post-doctoral stint at Stanford University. Dr. Roos’… Continue Reading









Leave a Reply