Talk Overview
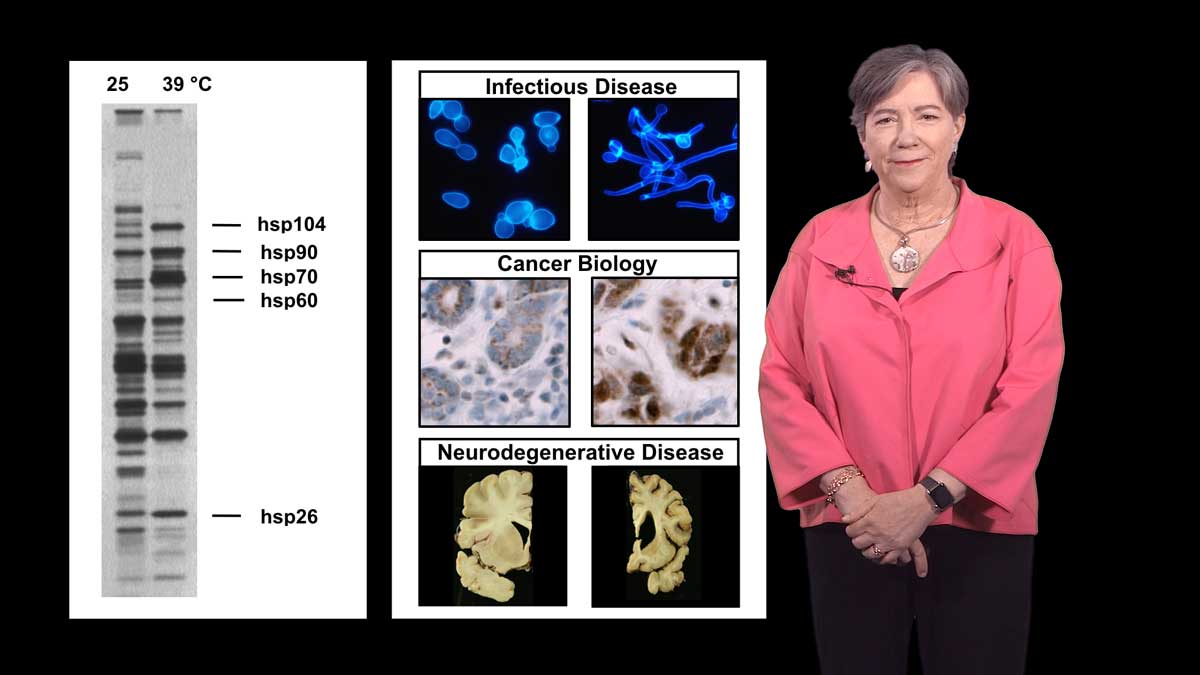
Dr. Petsko begins his lecture by presenting the challenges associated with a growing elderly population and a shrinking work force. As the population ages, we face an epidemic of debilitating neurodegenerative disease that will take a great financial and emotional toll on family, caregivers and society. The brains of patients with Alzheimer’s, Parkinson’s, and ALS/Lou Gehrig’s diseases are characterized by the presence of protein aggregates due to protein misfolding. While most neurodegenerative disease arises sporadically, about 10% has a direct genetic cause. Petsko explains that by studying the familiar forms, scientists have gained great insight into the cellular and molecular processes underlying these devastating diseases.
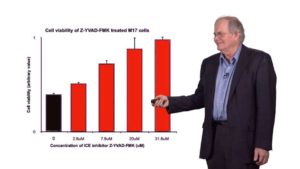
In Part 2, Petsko focuses on Parkinson’s disease, the second most common neurodegenerative disease after Alzheimer’s. Petsko and his colleagues studied patients with a genetic predisposition to Parkinson’s disease and found a mutation in the α-synuclein gene that caused the protein to misfold and aggregate. In sporadically occurring cases of Parkinson’s, they discovered that α-synuclein was cleaved and the resulting protein fragment formed aggregates. Switching to yeast as a model system and then to human cells, Petsko’s lab identified caspase-1 as the protease responsible for cleaving α-synuclein. Interestingly, caspase-1 is activated during inflammation, providing a possible explanation for how head injury and brain infection may contribute to Parkinson’s. The development of caspase-1 inhibitors that can penetrate the brain would present hope for an effective treatment for Parkinson’s disease.
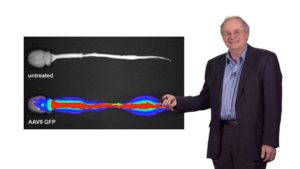
Petsko and others also studied patients with familial amyotrophic lateral sclerosis (ALS) or Lou Gehrig’s disease and he describes this work in Part 3. They knew that many of the genes mutated in ALS encode RNA-binding proteins and these proteins formed aggregates in neurons from ALS patients. Expression of two of these proteins, FUS and TDP43, in yeast resulted in the same phenotype. A screen for yeast genes that would suppress FUS/TDP43 toxicity identified five genes and all encoded RNA binding proteins. Excitingly, several of the human homologs of these genes also were shown to block FUS/TDP43 toxicity in human neuron and neonatal rat models. These encouraging results generate hope that targeted gene therapy may provide a future treatment for this terrible neurodegenerative disease.
Speaker Bio
Gregory Petsko

Gregory Petsko is Arthur J. Mahon Professor of Neuroscience in the Brain and Mind Research Institute at Weill Cornell Medical College. His lab studies protein structure and function with a particular focus on understanding and developing treatments or preventative therapies for age-related neurodegenerative diseases. Petsko received his B.A. from Princeton University. He was awarded a… Continue Reading





Reece Mia says
My mother was diagnosed with bulbar ALS in the summer of 2016;Her initial symptoms were quite noticeable. She first experienced weakness in her right arm and her speech and swallowing abilities were profoundly affected. She did little to seek help for this disease, as she had been her brother’s caregiver a few years earlier for the same disease.
She was able to get rid of the ideas with the help of totalcureherbsfoundation.com herbal formula that cured my Parkinson disease ,this herbal treatment helped my entire family during our worst time in life.
They exported worldwide and the herbal treatment will get to you in four days delivery to your address.
Tamra Palmer says
My husband was diagnosed with ALS (amyotrophic lateral sclerosis) when he was 63 years old 4 years ago. The Rilutek (riluzole) did very little to help him. The medical team did even less. His decline was rapid and devastating. His arms weakened first, then his hands and legs. Last year, a family friend told us about Organic Herbal Clinic and their successful ALS TREATMENT, we visited their website www. organicherbalclinic. com and ordered their ALS/MND Formula, i am happy to report the treatment effectively treated and reversed his Amyotrophic Lateral Sclerosis (ALS), most of the symptoms stopped, he is able to walk and able to ride his treadmill again, he is pretty active now.