Talk Overview
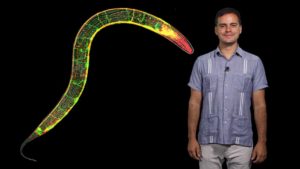
A fundamental question in neuroscience is how synapses are assembled in living animals to produce behaviors and store memories. Dr. Daniel Colón-Ramos and his lab address these questions by studying the cell biology of the neuronal synapse. In the first part of his seminar series, he introduces approaches his group has pioneered and implemented to image and manipulate synapses in vivo and with single-cell resolution in the nematode C.elegans. He also discusses fundamental aspects of genetics, cell biology and neuroscience which are necessary for understanding his research program and, more generally, for understanding how scientists make use of model organisms to generate new knowledge.
In his second lecture, Dr. Colón-Ramos describes his group’s finding of a previously-unknown metabolic subcompartment that powers synaptic function. His group observed that under conditions of energy stress, glycolytic proteins, which are normally diffusely distributed in cells, co-localize to a punctate structure adjacent to synapses. The local formation of these complexes is required for the synaptic vesicle cycle and synaptic function. The discoveries contribute to an unsolved question in neuroscience: how local, rapid and transient changes in energy demands are met at synapses to sustain their function. He frames his lab’s cell biological findings in the context of the long history of research of energy metabolism, drawing new lessons and discussing how the discoveries now provide new opportunities to examine the cell biology of metabolism at the neuronal synapse.
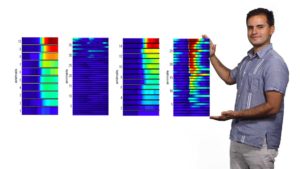
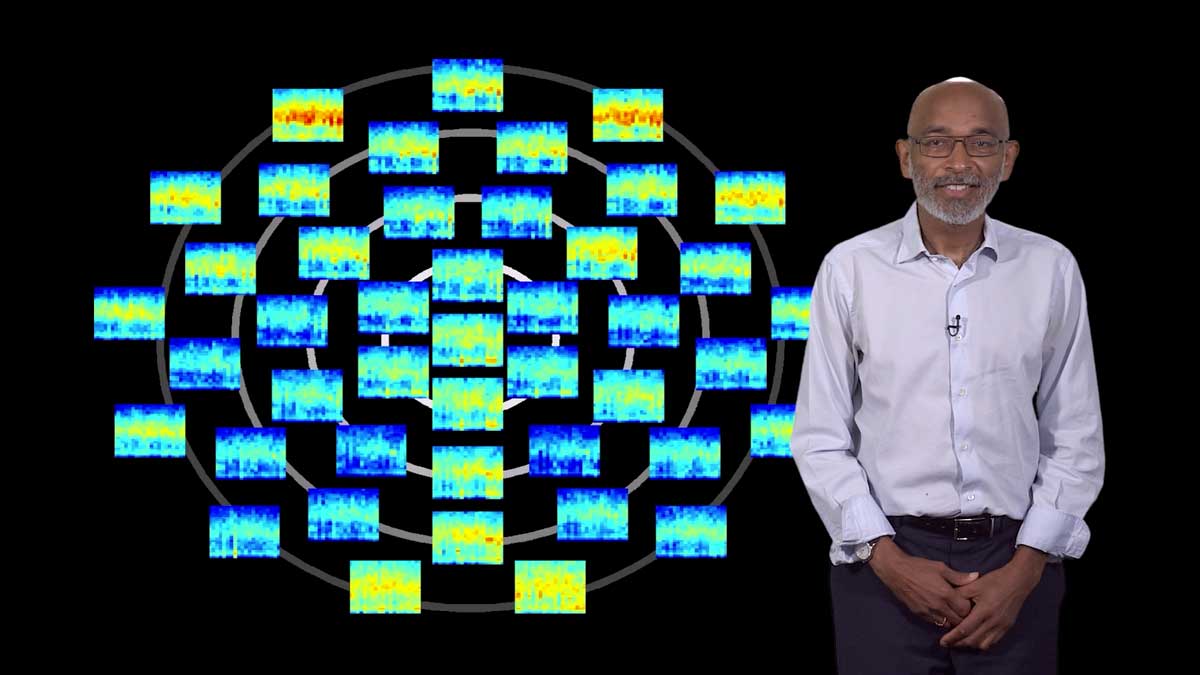
In his third and final lecture, Colón-Ramos discusses how his group uses the approaches they have developed to reduce system’s level questions, like behavior, to cell biological questions at the synapse. He describes his lab’s discovery that two plasticity mechanisms—sensory adaptation and presynaptic plasticity—act within a single cell in C. elegans to encode thermosensory information and actuate a temperature-preference memory. The integration of these plasticity mechanisms result in a single-cell logic system that can both represent sensory stimuli and guide memory-based behavioral preference. These findings allow the Colón-Ramos group to directly link cell biological changes at the neuronal synapse with memory and behavior.
Speaker Bio
Daniel Colon Ramos

Associate Professor of Neuroscience and Cell Biology; Associate Professor of Neuroscience
Yale School of Medicine Continue Reading








Leave a Reply